Pharmaceutical FDA Citation Trends, Now Instantly Actionable
Visualizing FDA Compliance Risk: Pharmaceutical Citation Trends.
A Look at the FDA’s Most-Cited Pharmaceutical Regulations—and What They Mean for Your Quality System
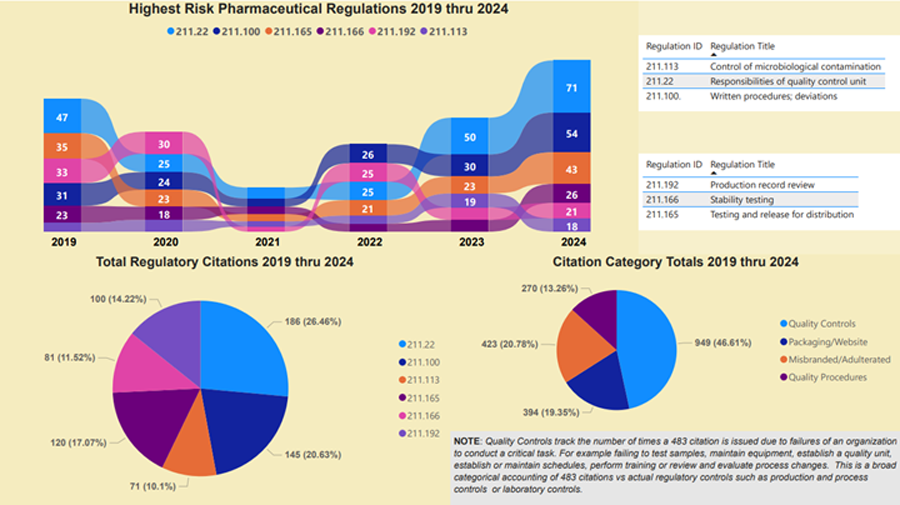
The data visualization above reveals six years of FDA citation activity tied to pharmaceutical CGMP violations—specifically the regulations that most frequently lead to 483 observations. It combines historical citation volume, regulation-level enforcement trends, and categorical risk analysis into one comprehensive view.
What you’re seeing:
Top Ribbon Chart : Tracks the six highest-risk 21 CFR 211 subparts cited from 2019 through 2024. These include:
- §211.22 – Responsibilities of the Quality Control Unit
- §211.100 – Written Procedures; Deviations
- §211.113 – Control of Microbiological Contamination
- §211.165 – Testing and Release for Distribution
- §211.166 – Stability Testing
- §211.192 – Production Record Review
- The spike in §211.22 and §211.113 citations in 2023–2024 signals a renewed FDA focus on quality oversight and microbial contamination control.
- Bottom Left Pie Chart: Breaks down total citation frequency by regulation, highlighting which specific CGMP sections were cited most often. §211.22 alone accounts for over 26% of all high-risk citations.
Bottom Right Pie Chart: Groups all FDA citations into four enforcement categories:
- Quality Controls (46.61%)
- Packaging/Website
- Misbranded/Adulterated
- Quality Procedures
This snapshot shows nearly half of all citations relate to failures in quality oversight—underscoring why quality unit accountability under §211.22 is so heavily scrutinized.
This dashboard empowers Regulatory Affairs and Quality Assurance professionals to identify trends, benchmark risks, and proactively strengthen internal controls where FDA attention is intensifying. Whether you’re facing upcoming inspections, planning your annual product review, or defending headcount during budget season, this tool helps you justify your strategy with evidence.
Want to explore this data filtered by your product class, facility type, or regulatory subpart?
We build fully customized versions of this dashboard for pharmaceutical manufacturers, CROs, and CMOs.
Let’s talk about what FDA compliance intelligence should look like for your team.
If You Are in Any of the Following Fields, This Dashboard Was Built for You
If you’re responsible for keeping your organization inspection-ready, this tool can help you proactively avoid Form 483 observations and FDA warning letters by pinpointing high-risk areas before they surface during an audit. This is not just a visualization—it’s a decision support system for FDA-regulated teams across the pharmaceutical supply chain.
This compliance intelligence dashboard is ideal for:
- Regulatory Affairs Professionals
Monitoring FDA trends, preparing submissions, and staying ahead of changing enforcement priorities. - Quality Assurance Managers & Directors
Identifying systemic risks, preparing for inspections, and driving CAPA decisions backed by data. - Compliance Officers
Overseeing site-wide or enterprise-level compliance programs and ensuring CGMP alignment. - Regulatory Intelligence & Surveillance Managers
Tracking real-time enforcement activity, benchmarking trends, and advising senior leadership. - Manufacturing Site Heads & Operations Managers
Benchmarking site performance against industry risk indicators and FDA citation patterns. - Pharmaceutical Consultants & Compliance Advisers
Providing insight-driven strategies to clients and staying ahead of regulatory hot spots. - Quality System Managers
Auditing processes, procedures, and SOPs to align with the most-cited CGMP sections. - VPs of Regulatory Affairs or Quality
Presenting inspection risk trends during management reviews and shaping long-term quality investments. - Risk Management Teams
Conducting compliance gap assessments and reducing exposure to enforcement actions. - GMP Training Leads
Using real citation data to tailor role-based training and reinforce high-risk SOPs. - CRO and CMO Leadership Teams
Proving oversight effectiveness and readiness to sponsors and regulators.
If you’re tired of retroactive compliance firefighting and want to prevent issues before the next FDA visit, this dashboard gives you the foresight your team needs.
ComplyFDA’s platform transforms inspection risk into clear, targeted action.
Let’s talk about a custom solution that aligns with your specific product class, site type, or global QA strategy.
Get a Custom Walkthrough of Your FDA Citation Risk Profile

